April 2025 Lab Outing

Introduction:
Research Topic
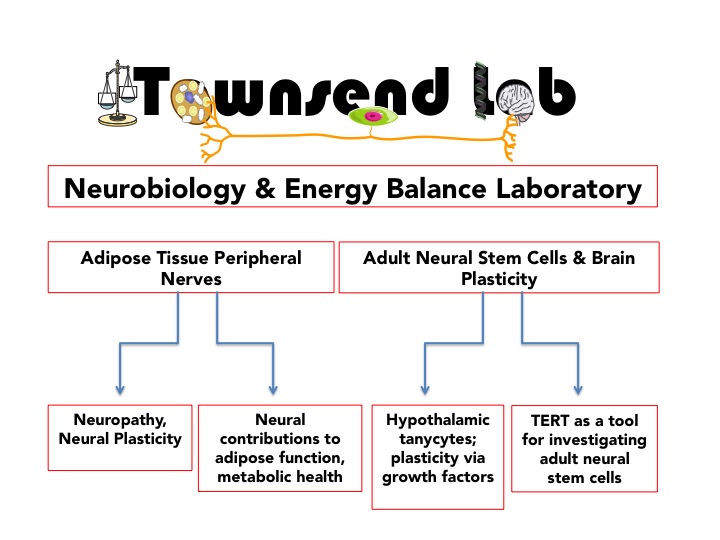
The Townsend Lab researches how the brain coordinates energy balance and how the central and peripheral nervous systems undergo plasticity in the control of energy balance processes like appetite and calorie burning. This research impacts the study of obesity, diabetes, metabolic syndrome/cardiometabolic diseases, aging, and neurodegenerative diseases including peripheral neuropathy. We utilize both in vitro and in vivo models, integrating the fields of neurobiology, metabolism & biochemistry, physiology & endocrinology, and cell/molecular biology.
Our work includes the following topics: obesity and body weight regulation; hypothalamic and extra-hypothalamic control of appetite; CNS control of fuel utilization and energetics; dietary effects on energy balance; control of adipose tissue development, metabolism, health, and functions (brown and white adipose tissues); activation of energy expenditure via peripheral nerve activity in adipose depots; adult neural stem cells; neurotrophic factors and neuropathy; and central and peripheral neural plasticity. In the adipose, we are focused on neuroimmune and neurovascular interactions.
Research Program
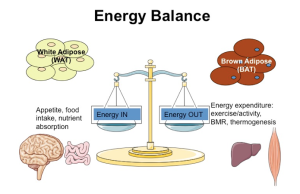
Research in the Townsend Lab focuses on the mechanisms by which the body maintains or loses energy balance, which is the balance between energy intake (comprised of dietary components, neural control of appetite, digestion and nutrient absorption) and energy expenditure (comprised of basal metabolism, mitochondrial function, thermogenesis, and physical activity). The brain, or central nervous system (CNS) is the part of the body responsible for coordinating and controlling these energy balance processes, and is in communication with other metabolic organs and tissues via a 2-way system that includes: neural connections with the brain (such as the sympathetic nerves innervating adipose depots) and endocrine products released from metabolic organs and tissues, which reach the brain through the systemic circulation. The over-arching goal of our research is to develop a deeper mechanistic understanding of how the body maintains energy balance, in order to prevent and treat metabolic diseases such as obesity and diabetes, which also disproportionately affect people in aging. As part of these energy balance processes, both central and peripheral nerves must undergo changes in plasticity, including neurogenesis, neurite outgrowth, and synaptogenesis. These pathways in the adult nervous system represent an intriguing mechanism by which energy balance may be controlled, and is another research focus in our lab.
Projects in the Townsend Lab fall under 2 broad themes:
Brain-Adipose Communication and Regulation of Peripheral Nerves in Adipose
For this project, we are interested in the bi-directional communication between the brain and both brown (energy-expending) and white (energy-storing) adipose tissues. This is accomplished through the endocrine system (circulating factors) and the nervous system. Adipose tissues are innervated by sympathetic nerves that send electrochemical signals from the brain, and release neurotransmitters like norepinephrine, which binds receptors on adipose tissue cell types. Adipose tissue also contains sensory nerves, which send electrical signals back to the brain and release neuropeptides to the tissue. We are interested in further identifying the neurotransmitters, neuropeptides, and neurotrophic factors that are active in adipose tissue and their functions in the tissue to maintain metabolic health, as well as learning more about how adipose nerve plasticity is regulated versus mechanisms of obesity/diabetes or age-related peripheral neuropathy. Research has already revealed the importance of adipose tissue neural innervation, as loss of this nerve supply through chemical, surgical or genetic denervation techniques leads to improper metabolic control in the tissue and may exacerbate conditions like obesity, metabolic syndrome, and diabetes. We were the first to identify ‘adipose neuropathy’ or the loss of adipose innervation, and the involvement of adipose Schwann cells, in states like obesity, diabetes and aging. Aspects of this project include neurovascular and neuroimmune interactions in adipose tissue.
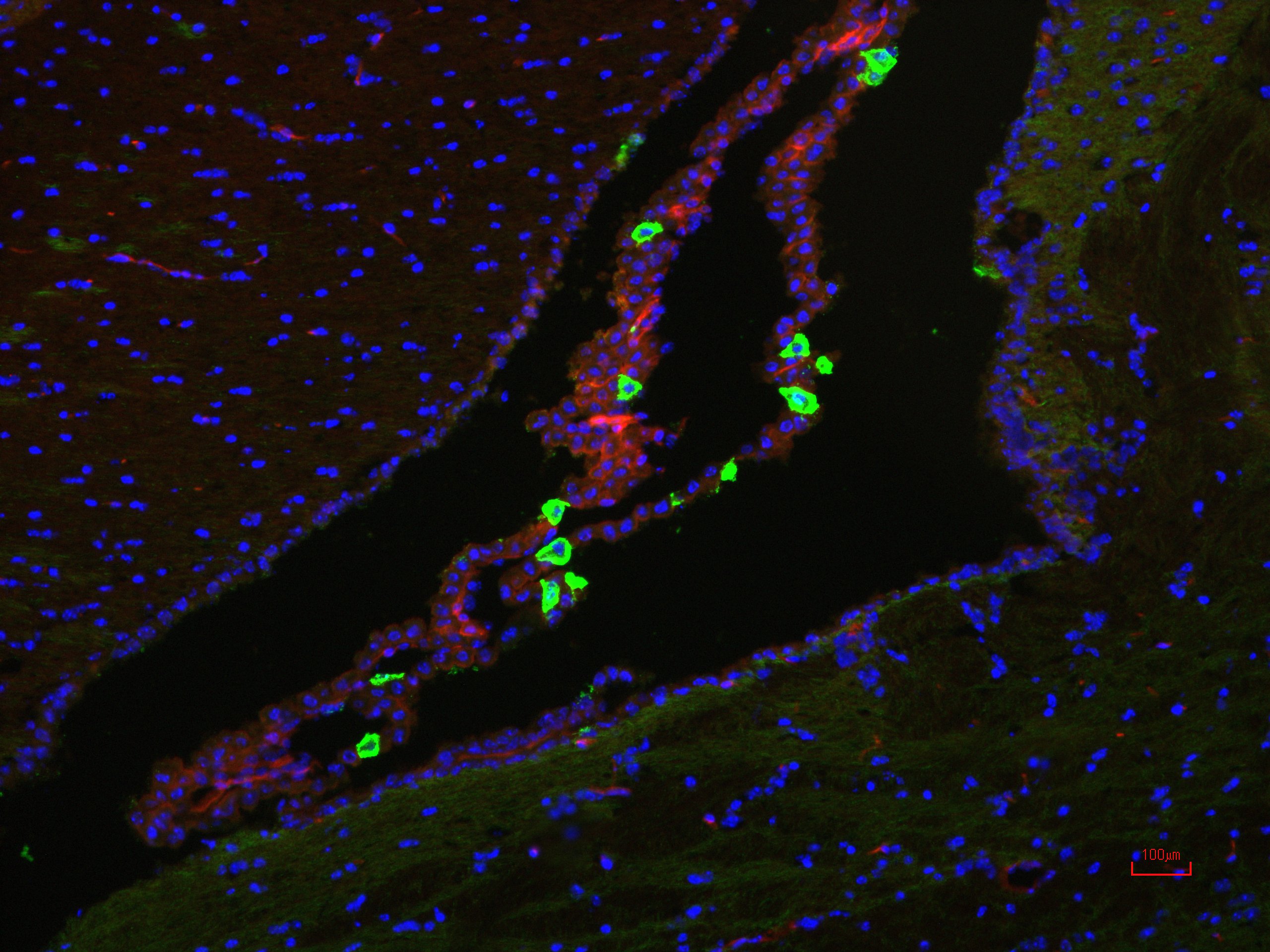
Adult Brain Plasticity and Hypothalamic Adult Stem Cells in the Control of Energy Balance
For this project, we are investigating novel populations of adult stem cells in the brain and determining how metabolic state (ie: exercise, fasting/re-feeding, diet, aging) impacts the ability of these cells to proliferate, differentiate, migrate, and integrate in the mature circuitry of the brain. Hypothalamic tanycytes are one population of cells known to be involved with both adult neural plasticity and metabolic control. In addition, we are exploring the involvement of the bone morphogenetic proteins (BMPs), a family of growth factors that we have demonstrated are important for regulating energy balance in the brain. The BMPs are also likely involved in adult neural plasticity. Finally, we have identified telomerase reverse transcriptase (TERT) as a novel marker of putative adult brain stem cells that are quiescent.
Townsend Lab Sites:
University of Maine: Kristy Townsend